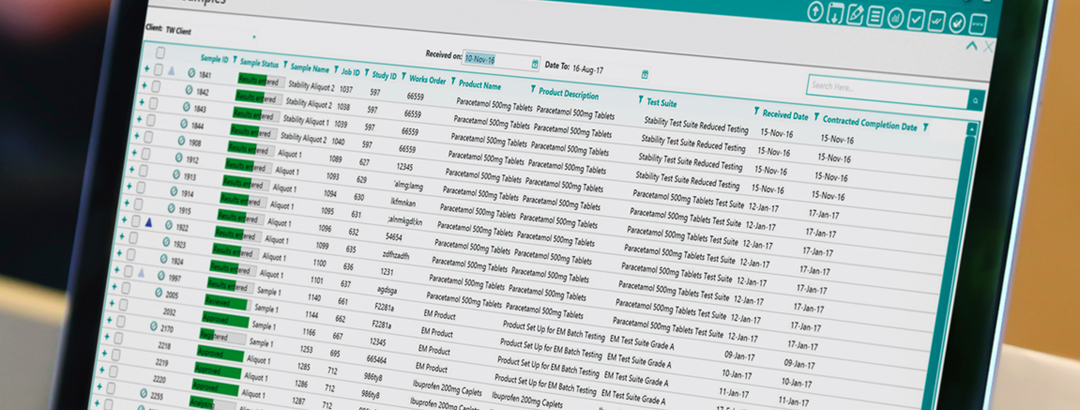
The key to a successful deployment of a LIMS in your laboratory is simple and it is to choose the LIMS provider that most suits the needs of your company. There are many LIMS providers to choose from and each with their own individual USP’s - the important thing is to find the one that is right for you. A LIMS is currently highly sought after by many laboratories as it can solve many of the issues that laboratories face such as time management, sample tracking and assisting in the gaining of accreditation. Indeed, many regulatory bodies would now expect a LIMS to be installed in certain laboratories as it is the most reliable method of data storage and ensuring the integrity of data. However, many laboratories are failing in their deployment of LIMS projects and these are just a few of the reasons why:
Why Most Labs Fail at LIMS Deployment Projects
by Sophie Blackburn posted in LIMS, Project Management, best practices, deployment projects, Laboratory Efficiency, Laboratory Management, TipsContrary to Popular Belief, LIMS Doesn’t Have to be Complicated
by Sophie Blackburn posted in LIMS, Project Management, best practices, deployment projects, Laboratory Efficiency, Laboratory Management, TipsIf there is a word that you can say to most laboratory professionals to make them cringe, LIMS would be up there. Why has LIMS gained such a bad reputation in industry?
SaaS vs On-premise – Why Now is the Time to Consider a SaaS LIMS
by Sophie Blackburn posted in LIMS, Data Management, Process Improvement, best practices, process mapping, deployment projects, Laboratory Efficiency, Laboratory Management, TipsAdvancements in technology have allowed the availability and delivery of software to be quicker, more secure and more reliable. If you’re still using on-premise LIMS, here are a few reasons why you should consider switching to SaaS LIMS.
4 Reasons Why You Don't Have LIMS and Tips to Overcome Them
by Sophie Blackburn posted in LIMS, Data Management, Process Improvement, best practices, process mapping, deployment projects, Laboratory Efficiency, Laboratory Management, TipsLaboratory information management systems (LIMS) are known to deliver substantial efficiencies to laboratory operations. From sample management, data comparison and reporting, LIMS can transform the most time-consuming, mundane and critical tasks to simple, easy-to-complete tasks within a click of a button. So, why doesn’t every lab have a LIMS?
LabHQ's Best Features for Pharma
by Sophie Blackburn posted in LIMS, Data Management, Process Improvement, best practices, process mapping, deployment projects, Laboratory Efficiency, Laboratory Management, TipsThe pharmaceutical industry is highly regulated to ensure that the quality, efficacy and safety of medicines delivered to patients is maintained. When pharma companies
What to Do When Your LIMS is No Longer Supported
by Theresa Webster posted in LIMS, Data Management, Process Improvement, best practices, process mapping, deployment projects, Laboratory Efficiency, Laboratory Management, TipsAs a laboratory that has already made the leap from paper to LIMS, moving away from LIMS would be a step backwards, and certainly wouldn’t be the favourable option if your LIMS is no longer supported. What do you do when your LIMS supplier has discontinued service? In this blog, we go through the key areas to consider, so you don’t have to roll back to paper-based systems.
The Dont's of LIMS Project Management
by Vanessa Ford posted in LIMS, Project Management, deployment projectsLast week we issued our blog on the Do's of LIMS Project Management, and now that you're familiar with best practice, you can find here what we highlighted as NOT to do when managing a LIMS project.
The Do's of LIMS Project Management
by Vanessa Ford posted in LIMS, Project Management, deployment projectsAssign roles and responsibilities
Identifying the project team and its responsibilities, from Key Stakeholders to Ancillary Team Members, is one of the first steps in a project’s life. As a Project Manager you may need assistance from the Project Sponsor in identifying the appropriate personnel, and creation of a Stakeholder map may be useful.
I Know I Need LIMS But What Do I Do Now?
by Phil Saunders posted in LIMS, Data Management, Project Management, Process Improvement, LabHQ, process mapping, QC Laboratory Data Management, Quality, deployment projects